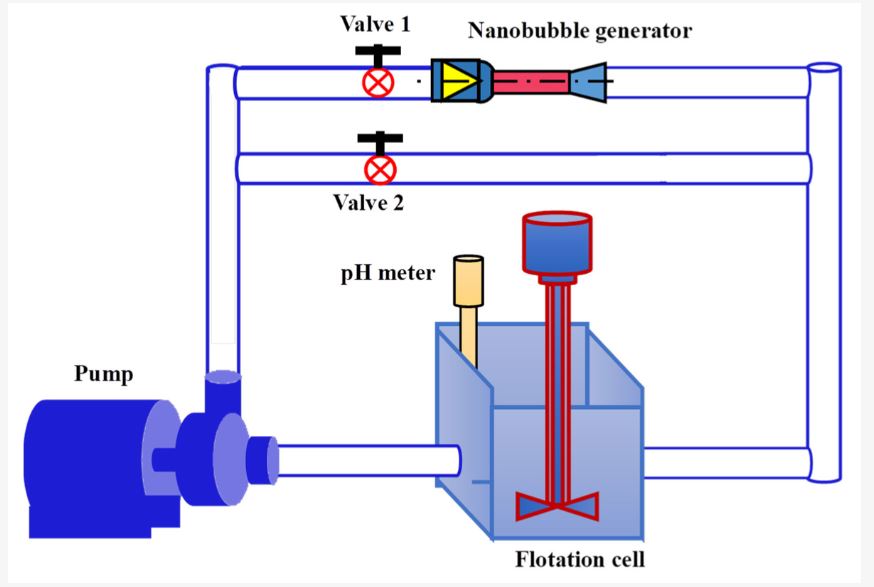
https://www.mdpi.com/2079-4991/12/19/3361#B31-nanomaterials-12-03361
This study was conducted to investigate the mechanisms of enhanced microfine
flake graphite (MFG) flotation by nanobubbles generated based on the principle
of hydrodynamic cavitation. The effects of nanobubbles on graphite surface
properties were characterized in terms of the flotation kinetics, collector adsorption
behavior, Zeta potential, IR spectra, contact angle, etc. The results show that the surface
nanobubbles increased the hydrophobic attraction and reduced the electrostatic
repulsion between the graphite particles and collector molecules, significantly improving
the flotation selectivity and the kinetic flotation rate and promoting the agglomeration of MFG.
.png)
.png)
Based on the above description and discussion of our experimental results on the
comparative flotation kinetics, adsorption capacity, Zeta potential, FTIR, and
contact angle with graphite particles under various conditions with and without
nanobubbles, the following conclusions can be drawn from this study:
(1)
The graphite flotation results showed that the flotation kinetics and the
rate of recovery and the grade of the concentrate were significantly enhanced
by the presence of nanobubbles;
(2)
The presence of the surface nanobubbles increased the adsorption rate and
capacity of diesel on the graphite surface, significantly improving its hydrophobicity.
The mineralization efficiency of the flotation process was also significantly
improved by the surface nanobubbles, which is partly responsible for the increased
graphite flotation kinetics and selectivity;
(3)
The nanobubbles formed on the surface of the graphite compound effectively
reduced the electrostatic repulsion between the graphite particles, promoting
the agglomeration of fine graphite particles and increasing the stability of the
graphite agglomerates. The surface nanobubbles also reduced the electrostatic
repulsion between the diesel droplets and graphite particles and increased the
adsorption capacity of diesel on the graphite surface, which improved the degree
of hydrophobicity of the graphite surface and the selectivity of flotation;
(4)
The FTIR results and contact angle measurements confirmed that the surface
nanobubbles improved the hydrophobicity of the graphite surface, increased the
hydrophobic attraction between the graphite particles and diesel droplets and
the adsorption capacity of diesel on the graphite surface, further improving the
degree of the hydrophobicity of the graphite surface;
(5)
Future studies are needed to investigate how nanobubbles function to mask the
hydrophilic sites on graphite surfaces. The interactions of nanobubbles with oil
droplets and the consequent effects on oil adsorption on graphite should also be
studied to achieve a better understanding of the fundamentals of nanobubble-enhanced flotation.